
Unusual Sulfur Chemistry to Build a Higher-Power Battery: and this isn’t just another flashy lab headline. This development represents a serious shift in how we may power everything from electric pickup trucks in Texas to rural microgrids in the Dakotas. Researchers are digging deep into unusual sulfur chemistry to create a higher-power battery that could outperform conventional lithium-ion technology while reducing dependence on expensive, imported materials. If you’ve been watching the battery space like I have over the years — from the early lithium-ion boom to today’s push toward solid-state — you know innovation usually comes in small steps. But every now and then, something different hits the scene. Sulfur-based battery chemistry isn’t new, but the way scientists are now stabilizing and controlling sulfur reactions is a fresh approach that could move this technology from theory to reality.
Before we dive deep, let’s frame the importance clearly: America’s energy transition depends heavily on reliable, affordable battery storage. And right now, lithium dominates that landscape. But lithium isn’t perfect. It’s costly, geopolitically sensitive, and environmentally demanding to extract. That’s why sulfur — an abundant and inexpensive material already produced in large quantities in the United States — is turning heads again.
Table of Contents
Unusual Sulfur Chemistry to Build a Higher-Power Battery
Scientists Tap Unusual Sulfur Chemistry to Build a Higher-Power Battery represents more than incremental progress. It reflects a broader shift toward energy independence, material innovation, and smarter chemistry design. Sulfur and sodium offer a compelling combination of abundance, affordability, and high theoretical performance. While challenges remain — particularly in durability and manufacturing scale — the progress being made suggests sulfur-based systems could play a major role in the next generation of American energy storage. From electric highways to resilient rural grids, this chemistry has serious potential. The science is advancing. The supply chain advantages are clear. And if researchers continue solving stability challenges, sulfur may soon power more than just industrial byproducts — it could power America’s future.
| Topic | Details |
|---|---|
| Breakthrough Focus | Unusual sulfur chemistry for higher-power batteries |
| Core Materials | Sulfur + Sodium (abundant and low-cost) |
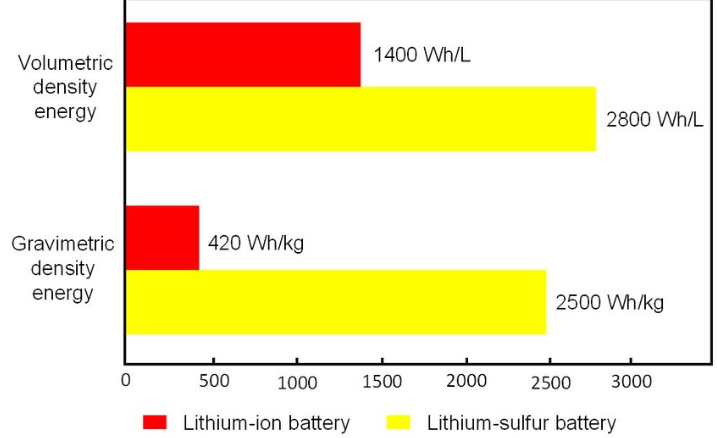
| Theoretical Energy Density | ~2,600 Wh/kg for lithium-sulfur (U.S. DOE) |
| Current Lithium-Ion Energy Density | ~250–300 Wh/kg |
| Major U.S. Sulfur Source | Petroleum refining byproduct |
| Market Impact Potential | EVs, grid storage, defense, aviation |
| Official Reference | https://www.energy.gov |
Why Sulfur Is Back in the Spotlight?
Let’s break this down in plain terms.
A battery stores energy through chemical reactions. In today’s lithium-ion batteries, lithium ions move between two electrodes through a liquid electrolyte. That movement generates power.
Sulfur-based batteries use sulfur as the cathode material. Sulfur can hold significantly more energy per unit weight than traditional lithium-ion cathodes. According to the U.S. Department of Energy (DOE), lithium-sulfur batteries have a theoretical energy density of around 2,600 watt-hours per kilogram, compared to roughly 250–300 Wh/kg for conventional lithium-ion batteries.
That’s not a small difference. That’s nearly a tenfold increase in potential.
But here’s the deal: theoretical performance isn’t the same as real-world performance.
Sulfur’s chemistry is complex. When sulfur reacts during charging and discharging, it forms compounds called polysulfides. These compounds dissolve into the electrolyte and migrate within the battery. That causes capacity loss, shortens lifespan, and reduces reliability.
For years, this “polysulfide shuttle effect” has held sulfur batteries back.
What’s different now? Scientists are approaching sulfur chemistry in a smarter way — stabilizing reactions rather than fighting them.
The Role of Sodium: A Strategic Shift
One of the most important aspects of this breakthrough is the use of sodium instead of lithium in some designs.
Why does that matter?
Lithium demand is projected to skyrocket. According to the International Energy Agency (IEA), demand for lithium could increase more than 40 times by 2040 under aggressive clean-energy scenarios. That kind of surge strains mining operations, increases costs, and introduces geopolitical vulnerabilities.
Sodium, on the other hand, is widely available. It’s derived from common salt (sodium chloride), and the United States has abundant access to it.
Switching to sodium-sulfur battery systems offers several advantages:
- Lower material cost
- Greater domestic supply security
- Reduced environmental extraction impact
- Improved scalability potential
From a national security standpoint, this matters. The U.S. government has emphasized domestic battery production under recent infrastructure and clean-energy initiatives. Reducing reliance on imported lithium strengthens supply chains.
How Unusual Sulfur Chemistry to Build a Higher-Power Battery Actually Works?
Let’s walk through the process step by step.
Rethinking the Cathode Structure
In traditional lithium-ion batteries, cathodes often contain materials like lithium cobalt oxide or nickel manganese cobalt (NMC). These materials are effective but expensive and resource-intensive.
Sulfur offers extremely high theoretical capacity — about 1,675 milliamp-hours per gram. That’s significantly higher than conventional cathode materials.
However, sulfur is electrically insulating. To function properly, it must be combined with conductive materials like carbon frameworks. Researchers are now engineering advanced porous carbon structures that hold sulfur in place and prevent it from dissolving into the electrolyte.
Controlling Chemical Reactions
Instead of allowing sulfur to freely form unstable intermediate compounds, researchers are designing electrolytes that chemically bind and stabilize sulfur species.
In some experimental systems, chlorine or alternative electrolyte additives help manage reaction pathways. By modifying the chemical environment, scientists reduce polysulfide migration and improve cycle life.
Improving Ion Transport
Efficient ion movement is key to power output. By optimizing the electrolyte and electrode interfaces, researchers can increase reaction speed, which directly improves power density.
Higher power density means the battery can deliver energy faster — crucial for applications like electric trucks accelerating onto highways or grid batteries responding to demand spikes.
Real-World Applications in the United States
This isn’t just academic research. The implications stretch across multiple industries.
Electric Vehicles
The U.S. electric vehicle market continues to grow. According to the U.S. Energy Information Administration (EIA), EV sales have reached record levels in recent years.
Higher energy density sulfur batteries could:
- Extend driving range beyond 500 miles
- Reduce battery weight
- Lower vehicle production costs
- Shorten charging times
For American automakers competing globally, that’s huge.
Imagine an electric pickup that can haul heavy loads across Montana without range anxiety. That’s the type of performance sulfur-based systems aim to enable.
Grid Energy Storage
Renewable energy sources like wind and solar are intermittent. The sun doesn’t always shine. The wind doesn’t always blow.
Affordable, high-capacity batteries are critical for grid stability.
Sulfur-based batteries could provide:
- Large-scale energy storage at lower cost
- Rural energy resilience
- Backup systems for critical infrastructure
The U.S. Department of Energy has emphasized grid modernization as a national priority. Cost-effective storage plays a major role.
Aerospace and Defense
Lightweight batteries matter in aviation and military applications. A higher energy density battery reduces payload weight and increases operational efficiency.
Sulfur’s lightweight nature makes it attractive for drones, satellites, and aircraft systems.
Unusual Sulfur Chemistry to Build a Higher-Power Battery: Challenges That Still Need Solving
Now let’s keep it honest.
Sulfur battery systems still face technical hurdles.
Cycle Life
Traditional lithium-ion batteries can exceed 1,000 charge cycles. Many sulfur-based systems historically struggled to maintain capacity beyond a few hundred cycles.
Researchers are working to improve structural stability and electrolyte chemistry to increase durability.
Safety
While sodium systems reduce certain lithium-related risks, any new chemistry must undergo extensive safety testing before commercial deployment.
Thermal stability, short-circuit resistance, and large-scale manufacturing safety protocols must be validated.
Manufacturing Infrastructure
Scaling lab breakthroughs into gigafactory production isn’t simple. New materials require new fabrication techniques, supply chains, and quality control systems.
Economic and Workforce Impact Of Unusual Sulfur Chemistry to Build a Higher-Power Battery
Battery manufacturing is a major job creator.
According to the Bureau of Labor Statistics (BLS), employment in architecture and engineering occupations is projected to grow steadily, especially in renewable energy and advanced manufacturing sectors.
Sulfur-based battery commercialization could drive:
- Materials science careers
- Chemical engineering roles
- Advanced manufacturing jobs
- Research and development positions
Universities across the U.S. are expanding energy storage research programs to meet workforce demand.
Environmental Considerations
Sulfur is typically produced as a byproduct of petroleum refining. Using it in batteries repurposes material that might otherwise go to industrial chemical applications.
Sodium extraction generally has lower environmental impact compared to lithium brine mining.
Reducing dependence on cobalt — often sourced from regions with labor and ethical concerns — also improves sustainability metrics.
That said, full lifecycle analysis must evaluate manufacturing, recycling, and disposal impacts before declaring sulfur systems fully “green.”
Practical Advice for Industry Professionals
If you’re in the battery or energy sector, here’s what to watch:
- Track pilot-scale sodium-sulfur demonstration projects
- Monitor DOE-funded research grants
- Evaluate compatibility with existing manufacturing lines
- Study electrolyte innovation trends
For investors, pay attention to startups focusing on sulfur cathode stabilization technologies.
For students, electrochemistry and materials science remain high-demand specialties.
Scientists Test Tea Growth Using Simulated Moon Soil
Breakthrough Imaging Lets Scientists Watch Electrons Move in Real Time
Ford Changes Course on Electric Vehicles With a New Lineup Plan
















